|
|
| Line 1: |
Line 1: |
| − | ==Contaminated Sediment Risk Assessment== | + | ==Estimating PCE/TCE Abiotic First-Order Reductive Dechlorination Rate Constants in Clayey Soils Under Anoxic Conditions== |
| − | [[Contaminated Sediments - Introduction | Contaminated sediments]] in rivers and streams, lakes, coastal harbors, and estuaries have the potential to pose ecological and human health risks. The goals of risk assessment applied to contaminated sediments are to characterize the nature and magnitude of the current and potential threats to human health, wildlife and ecosystem functioning posed by contamination; identify the key factors contributing to the potential health and ecological risks; evaluate how implementation of one or more remedy actions will mitigate the risks in the short and long term; and evaluate the risks and impacts from sediment management, both during and after any dredging or other remedy construction activities.
| + | The U.S. Department of Defense (DoD) faces many challenges in restoring aquifers at contaminated sites, often due to back-diffusion of tetrachloroethene (PCE) and trichloroethene (TCE) from low-permeability clay zones. The uptake, storage, and subsequent long-term release of these dissolved contaminants from clays are key processes in understanding the longevity, intensity, and risks associated with many persistent chlorinated ethene groundwater plumes. Although naturally occurring abiotic and biotic dechlorination processes in clays may reduce stored contaminant mass and significantly aid natural attenuation, no standardized field method currently exists to verify or quantify these reactions. It is critical to remediation design efforts to demonstrate and validate a cost-effective in situ approach for assessing these dechlorination processes using first-order rate constants. An approach was developed and applied across eight DoD sites to support Remedial Project Managers (RPMs) and regulators in evaluating natural attenuation potential in clay-rich environments. |
| | <div style="float:right;margin:0 0 2em 2em;">__TOC__</div> | | <div style="float:right;margin:0 0 2em 2em;">__TOC__</div> |
| | | | |
| | '''Related Article(s):''' | | '''Related Article(s):''' |
| − | *[[Contaminated Sediments - Introduction]]
| |
| − | *[[In Situ Treatment of Contaminated Sediments with Activated Carbon]]
| |
| − | *[[Sediment Capping]]
| |
| − | *[[Passive Sampling of Sediments]]
| |
| | | | |
| − | '''Contributor(s):'''
| + | *[[Monitored Natural Attenuation (MNA)]] |
| − | *Richard J. Wenning | + | *[[Monitored Natural Attenuation (MNA) of Chlorinated Solvents]] |
| − | *Sabine E. Apitz | + | *[[Monitored Natural Attenuation - Transitioning from Active Remedies]] |
| | + | *[[Matrix Diffusion]] |
| | + | *[[REMChlor - MD]] |
| | | | |
| − | '''Key Resource(s):''' | + | '''Contributors:''' Dani Tran, Dr. Charles Schaefer, Dr. Charles Werth |
| − | * Contaminated Sediment Remediation Guidance for Hazardous Waste Sites<ref name="USEPA2005">United States Environmental Protection Agency (USEPA), 2005. Contaminated Sediment Remediation Guidance for Hazardous Waste Sites. Office of Solid Waste and Emergency Response, Washington, D.C. EPA-540-R-05-012. OSWER 9355.0-85. Free download from: [https://semspub.epa.gov/work/HQ/174471.pdf USEPA] [[Media: EPA-540-R-05-012.pdf | Report.pdf]]</ref>
| |
| | | | |
| − | * Principles for Environmental Risk Assessment of the Sediment Compartment<ref name="Tarazona2014">Tarazona, J.V., Versonnen, B., Janssen, C., De Laender, F., Vangheluwe, M. and Knight, D., 2014. Principles for Environmental Risk Assessment of the Sediment Compartment: Proceedings of the Topical Scientific Workshop. 7-8 May 2013. European Chemicals Agency, Helsinki. Document ECHA-14-R-13-EN. Free download from: [https://echa.europa.eu/documents/10162/22816050/environmental_risk_assessment_final_en.pdf/3515b685-6601-40ce-bd48-3f8d5332c0f8 European Chemicals Agency] [[Media: ECHA-14-R-13-EN.pdf | Report.pdf]]</ref>
| + | '''Key Resource:''' |
| − | | + | *Schaefer, C.E, Tran, D., Nguyen, D., Latta, D.E., Werth, C.J., 2025. Evaluating Mineral and In Situ Indicators of Abiotic Dechlorination in Clayey Soils<ref name="SchaeferEtAl2025"/> |
| − | * Assessing and managing contaminated sediments: | |
| − | :: Part I, Developing an Effective Investigation and Risk Evaluation Strategy<ref name="Apitz2005a">Apitz, S.E., Davis, J.W., Finkelstein, K., Hohreiter, D.W., Hoke, R., Jensen, R.H., Jersak, J., Kirtay, V.J., Mack, E.E., Magar, V.S. and Moore, D., 2005. Assessing and Managing Contaminated Sediments: Part I, Developing an Effective Investigation and Risk Evaluation Strategy. Integrated Environmental Assessment and Management, 1(1), pp. 2-8. [https://doi.org/10.1897/IEAM_2004a-002.1 DOI: 10.1897/IEAM_2004a-002.1] Free access article from: [https://setac.onlinelibrary.wiley.com/doi/epdf/10.1897/IEAM_2004a-002.1 Society of Environmental Toxicology and Chemistry] [[Media: Apitz2005a.pdf | Report.pdf]]</ref>
| |
| − | :: Part II, Evaluating Risk and Monitoring Sediment Remedy Effectiveness<ref name="Apitz2005b">Apitz, S.E., Davis, J.W., Finkelstein, K., Hohreiter, D.W., Hoke, R., Jensen, R.H., Jersak, J., Kirtay, V.J., Mack, E.E., Magar, V.S. and Moore, D., 2005b. Assessing and Managing Contaminated Sediments: Part II, Evaluating Risk and Monitoring Sediment Remedy Effectiveness. Integrated Environmental Assessment and Management, 1(1), pp.e1-e14. [https://doi.org/10.1897/IEAM_2004a-002e.1 DOI: 10.1897/IEAM_2004a-002e.1]</ref>
| |
| | | | |
| | ==Introduction== | | ==Introduction== |
| − | Improving the management of [[Contaminated Sediments - Introduction | contaminated sediments]] is of growing concern globally. Sediment processes in both marine and freshwater environments are important to the function of aquatic ecosystems<ref name="Apitz2012">Apitz, S.E., 2012. Conceptualizing the role of sediment in sustaining ecosystem services: Sediment-Ecosystem Regional Assessment (SEcoRA), Science of the Total Environment, 415, pp. 9-30. [https://doi.org/10.1016/j.scitotenv.2011.05.060 DOI:10.1016/j.scitotenv.2011.05.060] Free download from: [https://d1wqtxts1xzle7.cloudfront.net/7588577/Apitz_SEcoRA%202012.pdf?1326618388=&response-content-disposition=inline%3B+filename%3DConceptualizing_the_role_of_sediment_in.pdf&Expires=1637094311&Signature=c2wczG59XxkitPjmBhc9PaODHJ8Vufg3gyzdG8tqGD6~mIVhLoz30E7eQNIghfMlH~jbch3KTVxMqD2AQFMQCSeXghIwqH~lXjGrEP07MJXCEgntzSW-V8Gws~33it5pEm9Ied64fSOvMLJR-PUXVr2OVTsVHQJHurHdGrtEmhUd90bKrC0NNlD28YLGQpkVUOlqa75e0K4sjPngwPUwUxhq18NAH6-1Uc3fQU5g5AjXwGph-VNe7EwzT-0do5OD056AsG-Eg8xIZi0ABJqMsg1wb92tIPpmmNy6ntdklHeN6tq~3IJFB7Tg8XYntQ-CGT8pYV9S7Kz14GhXVm9OQA__&Key-Pair-Id=APKAJLOHF5GGSLRBV4ZA Academia.edu]</ref>, and many organisms rely on certain sediment quality and quantity characteristics for their life cycle<ref name="Hauer2018">Hauer, C., Leitner, P., Unfer, G., Pulg, U., Habersack, H. and Graf, W., 2018. The Role of Sediment and Sediment Dynamics in the Aquatic Environment. In: Schmutz S., Sendzimir J. (ed.s) Riverine Ecosystem Management. Aquatic Ecology Series, vol. 8, pp. 151-169. Springer. [https://doi.org/10.1007/978-3-319-73250-3_8 DOI: 10.1007/978-3-319-73250-3_8] Open access book from: [https://library.oapen.org/bitstream/handle/20.500.12657/27726/1002280.pdf?seque#page=153 SpringerOpen]</ref>. Human health can also be affected by sediment conditions, either via direct contact, as a result of sediment impacts on water quality, or because of the strong influence sediments can have on the quality of fish and shellfish consumed by people<ref name="Greenfield2015">Greenfield, B.K., Melwani, A.R. and Bay, S.M., 2015. A Tiered Assessment Framework to Evaluate Human Health Risk of Contaminated Sediment. Integrated Environmental Assessment and Management, 11(3), pp. 459-473. [https://doi.org/10.1002/ieam.1610 DOI: 10.1002/ieam.1610]</ref>. A common approach to achieving the explicit management goals inherent in different sediment assessment frameworks in North America and elsewhere is the use of the ecological risk assessment (ERA)<ref name="USEPA1997a">US Environmental Protection Agency (USEPA), 1997. The Incidence and Severity of Sediment Contamination in Surface Waters of the United States: Volume 1, National Sediment Quality Survey. EPA-823R-97-006. Washington, DC. [[Media: EPA-823-R-97-006.pdf | Report.pdf]]</ref>. An ERA “evaluates the likelihood and magnitude of adverse effects from exposure to a chemical for organisms, such as animals, plants, or microbes, in the environment”<ref name="SETAC2018">Society of Environmental Toxicology and Chemistry (SETAC), 2018. Technical Issue Paper: Environmental Risk Assessment of Chemicals. SETAC, Pensacola, FL. 5 pp. Free download from: [https://cdn.ymaws.com/www.setac.org/resource/resmgr/publications_and_resources/setac_tip_era.pdf SETAC] [[Media: setac_tip_era2018.pdf | Report.pdf]]</ref>. An ERA provides information relevant to the management decision-making process<ref name="Stahl2001">Stahl, R.G., Bachman, R., Barton, A., Clark, J., deFur, P., Ells, S., Pittinger, C., Slimak, M., Wentsel, R., 2001. Risk Management: Ecological Risk-Based Decision Making. SETAC Press, Pensacola, FL, 222 pp. ISBN: 978-1-880611-26-5</ref>. It should be performed in a scientifically based, defensible manner that is cost-effective and protective of human health and the environment<ref name="CNO1999">Chief of Naval Operations (CNO), 1999. Navy Policy for Conducting Ecological Risk Assessments, Letter 5090, Ser N453E/9U595355, dated 05 April 99. Department of the Navy, Washington, DC. Free download from: [https://www.navfac.navy.mil/content/dam/navfac/Specialty%20Centers/Engineering%20and%20Expeditionary%20Warfare%20Center/Environmental/Restoration/er_pdfs/gpr/cno-ev-pol-era-19990405.pdf the US Navy] [[Media: CNO1999.pdf | Report.pdf]]</ref>. Therefore, science-based methods for assessing sediment quality and use of risk-based decision-making in sediment management are important for identifying conditions suspected to adversely affect ecological and human services provided by sediments, and predicting the likely consequences of different sediment management actions<ref name="Bridges2006">Bridges, T.S., Apitz, S.E., Evison, L., Keckler, K., Logan, M., Nadeau, S. and Wenning, R.J., 2006. Risk‐Based Decision Making to Manage Contaminated Sediments. Integrated Environmental Assessment and Management, 2(1), pp. 51-58. [https://doi.org/10.1002/ieam.5630020110 DOI: 10.1002/ieam.5630020110] Free access article from: [https://setac.onlinelibrary.wiley.com/doi/epdf/10.1002/ieam.5630020110 SETAC]</ref><ref name="Apitz2011">Apitz, S.E., 2011. Integrated Risk Assessments for the Management of Contaminated Sediments in Estuaries and Coastal Systems. In: Wolanski, E. and McLusky, D.S. (eds.) Treatise on Estuarine and Coastal Science, Vol 4, pp. 311–338. Waltham: Academic Press. ISBN: 9780123747112</ref>.
| + | Cost-effective methods are needed to verify the occurrence of natural dechlorination processes and quantify their dechlorination rates in clays under ambient in situ conditions in order to reliably predict their long-term influence on plume longevity and mass discharge. However, accurately determining these rates is challenging due to slow reaction kinetics, the transient nature of transformation products, and the interplay of biotic and abiotic mechanisms within the clay matrix or at clay-sand interfaces. Tools capable of quantifying these reactions and assessing their role in mitigating plume persistence would be a significant aid for long-term site management. |
| | | | |
| − | Sediment risk assessment is increasingly used by governmental agencies to support sediment management in freshwater, estuarine, and marine environments. Strategies for sediment management encompass a wide variety of actions, from removal, capping or treatment of contaminated sediment to the monitoring of natural processes, including sedimentation, binding, and bio- and photo-degradation that serve to reduce the potential threat to aquatic life over time. It is not uncommon to revisit a sediment risk assessment periodically to check how changed environmental conditions reflected in sediment and biotic sampling work has either reduced or exacerbated the threats identified in the initial assessment.
| + | For reductive abiotic dechlorination under anoxic conditions, a 1% hydrochloric acid (HCl) extraction of a sample of native clay coupled with X-ray diffraction (XRD) data can be used as a screening level tool to estimate reductive dechlorination rate constants. These rate constants can be inserted into fate and transport models such as [[REMChlor - MD]]<ref>Falta, R., and Wang, W., 2017. A semi-analytical method for simulating matrix diffusion in numerical transport models. Journal of Contaminant Hydrology, 197, pp. 39-49. [https://doi.org/10.1016/j.jconhyd.2016.12.007 doi: 10.1016/j.jconhyd.2016.12.007] [[Media: FaltaWang2017.pdf | Open Access Manuscript]]</ref><ref>Kulkarni, P.R., Adamson, D.T., Popovic, J., Newell, C.J., 2022. Modeling a well-charactized perfluorooctane sulfate (PFOS) source and plume using the REMChlor-MD model to account for matrix diffusion. Journal of Contaminant Hydrology, 247, Article 103986. [https://doi.org/10.1016/j.jconhyd.2022.103986 doi: 10.1016/j.jconhyd.2022.103986] [[Media: KulkarniEtAl2022.pdf | Open Access Manuscript]]</ref> to quantify abiotic dechlorination impacts within clay aquitards on chlorinated solvent plumes. Thus, determination of the abiotic reductive dechlorination rate constant for a particular clayey soil can be readily utilized to provide a more accurate assessment of aquifer cleanup timeframes for groundwater plumes that are being sustained by contaminant back-diffusion. |
| | | | |
| − | At present, several countries lack common recommendations specific to conducting risk assessment of contaminated sediments<ref name="Bruce2020">Bruce, P., Sobek, A., Ohlsson, Y. and Bradshaw, C., 2020. Risk assessments of contaminated sediments from the perspective of weight of evidence strategies – a Swedish case study. Human and Ecological Risk Assessment, 27(5), pp. 1366-1387. [https://doi.org/10.1080/10807039.2020.1848414 DOI: 10.1080/10807039.2020.1848414] [https://www.tandfonline.com/doi/full/10.1080/10807039.2020.1848414 Website]</ref>. In the European Union, sediment has played a secondary role in the Water Framework Directive (WFD), with most quality standards being focused on water with the option for the development of national standards for sediment and biota for bioaccumulative compounds. The Common Implementation Strategy (CIS) in 2010 provided guidance on the monitoring of contaminants in sediments and biota, but not on risk-based decision-making
| + | ==Recommended Approach== |
| | + | [[File: TranFig1.png | thumb | 500 px | Figure 1: First-order rate constants for abiotic reductive dechlorination of TCE under anaerobic conditions. Circles are data from Schaefer ''et al.'', 2021<ref>Schaefer, C.E., Ho, P., Berns, E., Werth, C., 2021. Abiotic dechlorination in the presence of ferrous minerals. Journal of Contaminant Hydrology, 241, 103839. [https://doi.org/10.1016/j.jconhyd.2021.103839 doi: 10.1016/j.jconhyd.2021.103839] [[Media: SchaeferEtAl2021.pdf | Open Access Manuscript]]</ref>, filled squares from Schaefer ''et al.'', 2018<ref name="SchaeferEtAl2018"/>, and Schaefer ''et al.'', 2017<ref>Schaefer, C.E., Ho., Gurr, C., Berns, E., Werth, C., 2017. Abiotic dechlorination of chlorinated ethenes in natural clayey soils: impacts of mineralogy and temperature. Journal of Contaminant Hydrology, 206, pp. 10-17. [https://doi.org/10.1016/j.jconhyd.2017.09.007 doi: 10.1016/j.jconhyd.2017.09.007] [[Media: SchaeferEtAl2017.pdf | Open Access Manuscript]]</ref>, and open squares from Schaefer ''et al.'', 2025<ref name="SchaeferEtAl2025"/>. ]] |
| | + | [[File: TranFig2.png | thumb | 600 px | Figure 2: Flowchart diagram of field screening procedures]] |
| | + | The recommended approach builds upon the methodology and findings of a recent study<ref name="SchaeferEtAl2025">Schaefer, C.E., Tran, D., Nguyen, D., Latta, D.E., Werth, C.J., 2025. Evaluating Mineral and In Situ Indicators of Abiotic Dechlorination in Clayey Soils. Groundwater Monitoring and Remediation, 45(2), pp. 31-39. [https://doi.org/10.1111/gwmr.12709 doi: 10.1111/gwmr.12709]</ref>, emphasizing field-based and analytical techniques to quantify abiotic first-order reductive dechlorination rate constants for PCE and TCE in clayey soils under anoxic conditions. Key components of this evaluation are listed below: |
| | + | #<u>Zone Identification:</u> The focus of the investigation should be to delineate clayey zones adjacent to hydraulically conductive zones. |
| | + | #<u>Ferrous Mineral Quantification:</u> Assess ferrous mineral context in clay via 1% HCl extraction at ambient temperature over a 10-minute interval. |
| | + | #<u>Mineralogical Characterization:</u> Conduct XRD analysis with the specific intent of identifying the presence of pyrite and biotite. |
| | + | #<u>Reduced Gas Analysis:</u> Measurement of reduced gases such as acetylene, ethene, and ethane concentrations in clay samples. Gas-tight sampling devices (e.g., En Core® soil samplers by En Novative Technologies, Inc.) should be used to ensure sample integrity during collection and transport. |
| | | | |
| − | ==Cap Design and Materials for Chemical Containment==
| + | Clay samples should be collected within a few centimeters of the high-permeability interface, with optional additional sampling further inward. For mineralogical analysis, a defined interval may be collected and subsequently subsampled. To preserve sample integrity, exposure to air should be minimized during collection, transport, and handling. Homogenization should occur within an anaerobic chamber, and if subsamples are required for external analysis, they must be shipped in gas-tight, anaerobic containers. |
| − | An inert material such as sand can be effective as a capping material where contaminants are strongly associated with solids and where the operative site specific transport mechanisms do not lead to rapid contaminant migration through such a material. Additional contaminant containment can often be achieved through the placement of clean sediment, e.g. dredged material from a nearby location. Other materials as cap layers or amendments may be useful to address particularly mobile contaminants or when particular degradative mechanisms can be exploited. The Anacostia River was the site of a demonstration that first tested “active” or “amended” capping in the field<ref name="Reible2003">Reible, D., Constant, D.W., Roberts, K. and Zhu, Y., 2003. Active capping demonstration project in anacostia DC. In Second International Conference on the Remediation of Contaminated Sediments: October. Free download available from: [https://www.researchgate.net/profile/Danny-Reible/publication/237747790_ACTIVE_CAPPING_DEMONSTRATION_PROJECT_IN_ANACOSTIA_DC/links/0c96053861030b7699000000/ACTIVE-CAPPING-DEMONSTRATION-PROJECT-IN-ANACOSTIA-DC.pdf ResearchGate]</ref><ref name="Reible2006">Reible, D., Lampert, D., Constant, D., Mutch Jr, R.D. and Zhu, Y., 2006. Active Capping Demonstration in the Anacostia River, Washington, DC. Remediation Journal: The Journal of Environmental Cleanup Costs, Technologies and Techniques, 17(1), pp. 39-53. [https://doi.org/10.1002/rem.20111 DOI: 10.1002/rem.20111] Free download available from: [https://www.academia.edu/download/44146457/Remediation_Journal_Paper_2006.pdf Academia.edu]</ref>. Amended caps are often the best option when groundwater upwelling or other advective processes promote significant mobility of contaminants and the addition of sorbents can slow that contaminant migration<ref name="Ghosh2011">Ghosh, U., Luthy, R.G., Cornelissen, G., Werner, D. and Menzie, C.A., 2011. In-situ Sorbent Amendments: A New Direction in Contaminated Sediment Management. Environmental Science and Technology, 45(4), pp. 1163-1168. [https://doi.org/10.1021/es102694h DOI: 10.1021/es102694h] Open access article from: [https://pubs.acs.org/doi/pdf/10.1021/es102694h American Chemical Society] [[Media: Ghosh2011.pdf | Report.pdf]]</ref>. Although a variety of materials have been proposed for sediment caps, a far smaller number of options have been successfully employed in the field.
| |
| − |
| |
| − | Metals migration is very site dependent due to the potential for many metals to complex with other species in the interstitial water and the specific metal speciation present at a site. Often, the strongly reducing environment beneath a cap renders many common metals unavailable through the formation of metal sulfides. In such cases, a simple sand cap can be very effective. Amended caps to manage metal contaminated sediments may be advantageous when site specific conditions lead to elevated metals mobility, but should be supported with site specific testing<ref name="Viana2008">Viana, P.Z., Yin, K. and Rockne, K.J., 2008. Modeling Active Capping Efficacy. 1. Metal and Organometal Contaminated Sediment Remediation. Environmental Science and Technology, 42(23), pp. 8922-8929. [https://doi.org/10.1021/es800942t DOI: 10.1021/es800942t]</ref>.
| |
| | | | |
| − | For hydrophobic organic contaminants, cap amendments that directly control groundwater upwelling and also sorbents that can remove migrating contaminants from that groundwater have been successfully employed. Examples include clay materials such as AquaBlok<sup>®</sup> for permeability control, sorbents such as [[Wikipedia: Activated carbon | activated carbon]] for truly dissolved contaminants, and [[Wikipedia: Organoclay | organophilic clays]] for separate phase contaminants.
| + | Estimation of the abiotic reductive first-order rate constant for PCE and TCE is based on the “reactive” ferrous content in the clay. Reactive ferrous content (Fe(II)<sub>r</sub>) is estimated as shown in Equation 1: |
| | | | |
| − | The placement of clean sediment as an ''in situ'' cap can be difficult when the material is fine grained or has a low density. Capping with a layer of coarse grained material such as clean sand mitigates this issue although clean sands have minimal sorption capacity. Because of this limitation, sand caps may not be sufficient for achieving remedial goals in sites where contamination levels are high or transport rates are fast due to pore water upwelling or tidal pumping effects. Conditions such as these may require the use of “active” amendments to reduce transport rates.
| + | ::'''Equation 1:''' <big>''Fe(II)<sub><small>r</small></sub> = DA + XRD<sub><small>pyr</small></sub> - XRD<sub><small>biotite</small></sub>''</big> |
| − |
| |
| − | Capping with clean sand provides a physical barrier between the underlying contaminated material and the overlying water, stabilizes the underlying sediment to prevent re-suspension of contaminated particles, and can reduce chemical exposure under certain conditions. Sand primarily provides a passive barrier to the downward penetration of bioturbating organisms and the upward movement of sediment or contaminants. Although conventional sandy caps can often be an effective means of managing contaminated sediments, there are conditions when sand caps may not be capable of achieving design objectives. Some factors that reduce the effectiveness of sand caps include:
| |
| | | | |
| − | *erosion and loss of cap integrity
| + | where ''DA'' is the ferrous content from the dilute acid (1% HCl) extraction, ''XRD<sub><small>pyr</small></sub>'' is the pyrite content from XRD analysis, and ''XRD<sub><small>biotite</small></sub>'' is the biotite content from XRD analysis<ref name="SchaeferEtAl2025"/>. |
| − | *high groundwater upwelling rates
| |
| − | *mobile (low sorption) contaminants of concern (COCs)
| |
| − | *high COC concentrations
| |
| − | *unusually toxic COCs
| |
| − | *the presence of tidal influences
| |
| − | *the presence of non-aqueous phase liquids (NAPLs)
| |
| − | *high rates of gas ebullition
| |
| | | | |
| − | Of these, the first three are common limitations to capping and often control the ability to effectively design and implement a cap as a sediment remedial strategy. In these cases, it may be possible to offset these issues by increasing the thickness of the cap. However, the required thickness can reach infeasible levels in shallow streams or navigable water bodies. In addition, increased construction costs associated with thick caps may become prohibitive. As a result of these issues, caps that use alternative materials (also known as active caps) to reduce the thickness or increase the protectiveness of a cap may be necessary. The materials in active caps are designed to interact with the COCs to enhance the containment properties of the cap.
| + | Abiotic dechlorination is unlikely to contribute to mitigating contaminant back-diffusion when reactive ferrous iron (Fe(II)<sub><small>r</small></sub>) concentrations are below 100 mg/kg (Figure 1). For Fe(II)<sub><small>r</small></sub> above 100 mg/kg, the first-order rate constant for PCE and TCE reductive dechlorination can be estimated using the correlation shown in Figure 1<ref name="SchaeferEtAl2018">Schaefer, C.E., Ho, P., Berns, E., Werth, C., 2018. Mechanisms for abiotic dechlorination of trichloroethene by ferrous minerals under oxic and anoxic conditions in natural sediments. Environmental Science and Technology, 52(23), pp.13747-13755. [https://doi.org/10.1021/acs.est.8b04108 doi: 10.1021/acs.est.8b04108]</ref><ref>Borden, R.C., Cha, K.Y., 2021. Evaluating the impact of back diffusion on groundwater cleanup time. Journal of Contaminant Hydrology, 243, Article 103889. [https://doi.org/10.1016/j.jconhyd.2021.103889 doi: 10.1016/j.jconhyd.2021] [[Media: BordenCha2021.pdf | Open Access Manuscript]]</ref>. The rate constant exhibits a strong positive correlation with the logarithm of reactive Fe(II) content (Pearson’s ''r'' = 0.82), with a slope of 4.7 × 10⁻⁸ L g⁻¹ d⁻¹ (log mg kg⁻¹)⁻¹. |
| | | | |
| − | [[Wikipedia: Apatite | Apatites]] are a class of naturally occurring minerals that have been investigated as a sorbent for metals in soils and sediments<ref name="Melton2003">Melton, J.S., Crannell, B.S., Eighmy, T.T., Wilson, C. and Reible, D.D., 2003. Field Trial of the UNH Phosphate-Based Reactive Barrier Capping System for the Anacostia River. EPA Grant R819165-01-0</ref><ref name="Reible2003"/><ref name="Knox2012">Knox, A.S., Paller, M.H. and Roberts, J., 2012. Active Capping Technology—New Approaches for In Situ Remediation of Contaminated Sediments. Remediation Journal, 22(2), pp.93-117. [https://doi.org/10.1002/rem.21313 DOI: 10.1002/rem.21313] Free download available from: [https://www.researchgate.net/profile/Anna-Knox-2/publication/233374607_Active_Capping_Technology-New_Approaches_for_In_Situ_Remediation_of_Contaminated_Sediments/links/5a7de4c5aca272a73765c344/Active-Capping-Technology-New-Approaches-for-In-Situ-Remediation-of-Contaminated-Sediments.pdf ResearchGate]</ref>. Apatites consist of a matrix of calcium phosphate and various other common anions, including fluoride, chloride, hydroxide, and occasionally carbonate. Metals are sequestered either through direct ion exchange with the calcium atom or dissolution of hydroxyapatite followed by precipitation of lead apatite. [[Wikipedia: Zeolite | Zeolites]], which are microporous aluminosilicate minerals with a high cationic exchange capacity (CEC), have also been proposed to manage metal species<ref name="Zhan2019">Zhan, Y., Yu, Y., Lin, J., Wu, X., Wang, Y. and Zhao, Y., 2019. Simultaneous control of nitrogen and phosphorus release from sediments using iron-modified zeolite as capping and amendment materials. Journal of Environmental Management, 249, p.109369. [https://doi.org/10.1016/j.jenvman.2019.109369 DOI: 10.1016/j.jenvman.2019.109369]</ref>.
| + | Figure 2 presents a decision flowchart designed to evaluate the significance and extent of abiotic reductive dechlorination. By applying Equation 1 to the dilute acid extractable Fe(II) plus measured mineral species data from clay samples, the reactive ferrous iron content (Fe(II)<sub><small>r</small></sub>) can be quantified, enabling a streamlined assessment of the extent to which abiotic processes are contributing to the mitigation of contaminant back-diffusion. |
| − |
| |
| − | It is possible to create a hydrophobic, sorbing layer for non-polar organics by exchanging a cationic surfactant onto the surface of clays such as zeolites and bentonites,. Organoclay is a modified bentonite containing such substitutions that has been evaluated for control of non-aqueous phase NAPLs and other organic contaminants<ref name="Reible2007">Reible, D.D., Lu, X., Moretti, L., Galjour, J. and Ma, X., 2007. Organoclays for the capping of contaminated sediments. AIChE Annual Meeting. ISBN: 978-081691022-9</ref>. An organoclay cap has been implemented for sediment remediation at the McCormick and Baxter site in Portland, OR<ref name="Parrett2005">Parrett, K. and Blishke, H., 2005. 23-Acre Multilayer Sediment Cap in Dynamic Riverine Environment Using Organoclay an Adsorptive Capping Material. Presentation to Society of Environmental Toxicology and Chemistry (SETAC), 26th Annual Meeting.</ref>. A similar organic sorbing phase can be formed by treating zeolites with surfactants but this approach has not been reported for contaminated sediments.
| |
| | | | |
| − | Activated carbon is a strong sorbent of hydrophobic organic compounds and has been used as a [[In Situ Treatment of Contaminated Sediments with Activated Carbon | treatment for sediments]] or as an active sorbent within a capping layer<ref name="Zimmerman2004">Zimmerman, J.R., Ghosh, U., Millward, R.N., Bridges, T.S. and Luthy, R.G., 2004. Addition of Carbon Sorbents to Reduce PCB and PAH Bioavailability in Marine Sediments: Physicochemical Tests. Environmental Science and Technology, 38(20), pp. 5458-5464. [https://doi.org/10.1021/es034992v DOI: 10.1021/es034992v]</ref><ref name="Werner2005">Werner, D., Higgins, C.P. and Luthy, R.G., 2005. The sequestration of PCBs in Lake Hartwell sediment with activated carbon. Water Research, 39(10), pp. 2105-2113. [https://doi.org/10.1016/j.watres.2005.03.019 DOI: 10.1016/j.watres.2005.03.019]</ref><ref name="Abel2018">Abel, S. and Akkanen, J., 2018. A Combined Field and Laboratory Study on Activated Carbon-Based Thin Layer Capping in a PCB-Contaminated Boreal Lake. Environmental Science and Technology, 52(8), pp. 4702-4710. [https://doi.org/10.1021/acs.est.7b05114 DOI: 10.1021/acs.est.7b05114] Open access article available from: [https://pubs.acs.org/doi/pdf/10.1021/acs.est.7b05114 American Chemical Society] [[Media: Abel2018.pdf | Report.pdf]]</ref><ref name="Payne 2018">Payne, R.B., Ghosh, U., May, H.D., Marshall, C.W. and Sowers, K.R., 2019. A Pilot-Scale Field Study: In Situ Treatment of PCB-Impacted Sediments with Bioamended Activated Carbon. Environmental Science and Technology, 53(5), pp. 2626-2634. [https://doi.org/10.1021/acs.est.8b05019 DOI: 10.1021/acs.est.8b05019]</ref><ref name="Yan2020">Yan, S., Rakowska, M., Shen, X., Himmer, T., Irvine, C., Zajac-Fay, R., Eby, J., Janda, D., Ohannessian, S. and Reible, D.D., 2020. Bioavailability Assessment in Activated Carbon Treated Coastal Sediment with In situ and Ex situ Porewater Measurements. Water Research, 185, p. 116259. [https://doi.org/10.1016/j.watres.2020.116259 DOI: 10.1016/j.watres.2020.116259]</ref>. Placement of activated carbon for sediment capping is difficult due to the near neutral buoyancy of the material but it has been applied in this manner in relatively low energy environments such as Onondaga Lake, Syracuse, NY<ref name="Vlassopoulos2017">Vlassopoulos, D., Russell, K., Larosa, P., Brown, R., Mohan, R., Glaza, E., Drachenberg, T., Reible, D., Hague, W., McAuliffe, J. and Miller, S., 2017. Evaluation, Design, and Construction of Amended Reactive Caps to Restore Onondaga Lake, Syracuse, New York, USA. Journal of Marine Environmental Engineering, 10(1), pp. 13-27. Free download available from: [https://www.researchgate.net/publication/317762995_Evaluation_design_and_construction_of_amended_reactive_caps_to_restore_Onondaga_lake_Syracuse_New_York_USA ResearchGate]</ref>. Alternatives in higher energy environments include placement of activated carbon in a mat such as the CETCO Reactive Core Mat (RCM)<sup>®</sup> or Huesker Tektoseal<sup>®</sup>, or as a composite material such as SediMite<sup>®</sup> or AquaGate<sup>®</sup>. In the case of the mats, powdered or granular activated carbon can be placed in a controlled layer while the density of the composite materials is such that they can be broadcast from the surface and allowed to settle to the bottom. In a sediment treatment application, the composite material would either be worked into the surface or allowed to intermix gradually by bioturbation and other processes. In a capping application, the mat or composite material would typically be combined or overlain with a sand or other capping layer to keep it in place and to provide a chemical isolation layer away from the sediment surface.
| + | If Fe(II)r is ≥ 100 mg/kg, a first-order dechlorination rate constant can be estimated and subsequently used within a contaminant fate and transport model. However, if acetylene is detected in the clay, even with Fe(II)r less than 100 mg/kg, then bench-scale testing using methods similar to those described in a recent study<ref name="SchaeferEtAl2025"/> is recommended, as such results would likely be inconsistent with those shown in Figure 1, suggesting some other mechanism might be involved, or that the system mineralogy might be more complex than anticipated. Even if Fe(II)r ≥ 100 mg/kg, confirmatory bench-scale testing may be conducted for additional verification and to refine estimation of the abiotic dechlorination rate constant. |
| | | | |
| − | As an alternative to a sorptive capping amendment, low-permeability cap amendments have been proposed to enhance cap design life by decreasing pore water advection. Low permeability clays are an effective means to divert upwelling groundwater away from a contaminated sediment area but are difficult to place in the aqueous environment. Bentonite clays can be placed in mats similar to what is done to provide a low permeability liner in landfills. There are also commercial products that can place clays directly such as the composite material AquaBlok<sup>®</sup>, a bentonite clay and polymer based mineral around an aggregate core<ref name="Barth2008">Barth, E.F., Reible, D. and Bullard, A., 2008. Evaluation of the physical stability, groundwater seepage control, and faunal changes associated with an AquaBlok<sup>®</sup> sediment cap. Remediation: The Journal of Environmental Cleanup Costs, Technologies and Techniques, 18(4), pp.63-70. [https://doi.org/10.1002/rem.20183 DOI: 10.1002/rem.20183]</ref>.
| + | ==Summary and Recommendations== |
| − |
| + | The approach outlined above is intended to serve as a generalized guide for practitioners and site managers to cost-effectively determine the extent to which beneficial abiotic reductive dechlorination reactions are likely occurring in low permeability (e.g., clayey) zones. This approach may be contraindicated if co-contaminants are present. It is currently unclear whether other classes of potentially reactive chemicals, such as trinitrotoluene (TNT) or chlorinated ethanes, could interact competitively with PCE and TCE. |
| − | Sediment caps become colonized by microorganisms from the sediments and surface water and potentially become a zone of pollutant biotransformation over time. Aerobic degradation occurs only near the solids-water interface in which benthic organisms are active and thus there might still be significant benthic organism exposure to contaminants. Biotransformation in the anaerobic zone of a cap, which typically extends well beyond the zone of benthic activity, could significantly reduce the risk of pollutant exposure but successful caps encouraging deep degradation processes have not been demonstrated beyond the laboratory. The addition of materials such as nutrients and oxygen releasing compounds for enhancing the attenuation of contaminants through biodegradation has also been assessed but not applied in the field. Short term improvements in biodegradation rates can be achieved through tailoring of conditions or addition of nutrients but long term efficacy has not been demonstrated<ref name="Pagnozzi2020">Pagnozzi, G., Carroll, S., Reible, D.D. and Millerick, K., 2020. Biological Natural Attenuation and Contaminant Oxidation in Sediment Caps: Recent Advances and Future Opportunities. Current Pollution Reports, pp.1-14. [https://doi.org/10.1007/s40726-020-00153-5 DOI: 10.1007/s40726-020-00153-5]</ref>.
| |
| − | [[File: SedCapFig2.png | thumb |600px|Figure 2. A conceptualization of a cap with accompanying habitat layer]]
| |
| | | | |
| − | ==Cap Design and Materials for Habitat Restoration==
| + | In addition, it remains unclear how other classes of compounds such as per- and polyfluoroalkyl substances (PFAS) may interact or sorb with ferrous minerals and potentially inhibit abiotic dechlorination reactions. Coupling these recommended activities with conventional site investigation tasks would provide an opportunity to perform many of the up-front screening activities with minimal additional project costs. It is important to note that the guidance proposed herein pertains to particularly low permeability media. Sites with complex or varying lithology, where the mineralogy and/or redox conditions may vary, might require evaluation of multiple samples to provide appropriate site-wide information. |
| − | In addition to providing chemical isolation and containment, a cap can also be used to provide improvements for organisms by enhancing the habitat characteristics of the bottom substrate<ref name="Yozzo2004">Yozzo, D.J., Wilber, P. and Will, R.J., 2004. Beneficial use of dredged material for habitat creation, enhancement, and restoration in New York–New Jersey Harbor. Journal of Environmental Management, 73(1), pp. 39-52. [https://doi.org/10.1016/j.jenvman.2004.05.008 DOI: 10.1016/j.jenvman.2004.05.008]</ref><ref name="Zhang2016">Zhang, C., Zhu, M.Y., Zeng, G.M., Yu, Z.G., Cui, F., Yang, Z.Z. and Shen, L.Q., 2016. Active capping technology: a new environmental remediation of contaminated sediment. Environmental Science and Pollution Research, 23(5), pp.4370-4386. [https://doi.org/10.1007/s11356-016-6076-8 DOI: 10.1007/s11356-016-6076-8]</ref><ref name="Vlassopoulos2017"/>. Often, contaminated sediment environments are degraded for a variety of reasons in addition to the toxic constituents. One way to overcome this is to provide both a habitat layer and chemical isolation or contaminant capping layer. Figure 2 illustrates just such a design providing a more appropriate habitat enhancing substrate, in this case by incorporation additional organic material, vegetation and debris, which is often used by fish species for protection, into the surface layer. In a high energy environment, it should be recognized that it may not be possible to keep a suitable habitat layer in place during high flow events. This would be true of suitable habitat that had developed naturally as well as a constructed habitat layer and it is presumed that if such a habitat is the normal condition of the waterbody that it will recover over time between such high flow events. | |
| | | | |
| − | ==Summary== | + | <br clear="right"/> |
| − | Clean substrate can be placed at the sediment-water interface for the purposes of reducing exposure to and risk from contaminants in the sediments. The cap can consist of simple materials such as sand designed to physically stabilize contaminated sediments and separate the benthic community from those contaminants or may include other materials designed to sequester contaminants even under adverse conditions including strong groundwater upwelling or highly mobile contaminants. The surface of a cap may be designed of coarse material such as gravel or cobble to be stable under high flow events or designed to be more appropriate habitat for benthic and aquatic organisms. As a result of its flexibility, simplicity and low cost relative to its effectiveness, capping is one of the most prevalent remedial technologies for sediments.
| |
| | | | |
| | ==References== | | ==References== |
| Line 72: |
Line 55: |
| | | | |
| | ==See Also== | | ==See Also== |
| | + | *[https://serdp-estcp.mil/projects/details/a7e3f7b5-ed82-4591-adaa-6196ff33dd60 ESTCP Project ER20-5031 – In Situ Verification and Quantification of Naturally Occurring Dechlorination Rates in Clays: Demonstrating Processes that Mitigate Back-Diffusion and Plume Persistence] |
Estimating PCE/TCE Abiotic First-Order Reductive Dechlorination Rate Constants in Clayey Soils Under Anoxic Conditions
The U.S. Department of Defense (DoD) faces many challenges in restoring aquifers at contaminated sites, often due to back-diffusion of tetrachloroethene (PCE) and trichloroethene (TCE) from low-permeability clay zones. The uptake, storage, and subsequent long-term release of these dissolved contaminants from clays are key processes in understanding the longevity, intensity, and risks associated with many persistent chlorinated ethene groundwater plumes. Although naturally occurring abiotic and biotic dechlorination processes in clays may reduce stored contaminant mass and significantly aid natural attenuation, no standardized field method currently exists to verify or quantify these reactions. It is critical to remediation design efforts to demonstrate and validate a cost-effective in situ approach for assessing these dechlorination processes using first-order rate constants. An approach was developed and applied across eight DoD sites to support Remedial Project Managers (RPMs) and regulators in evaluating natural attenuation potential in clay-rich environments.
Related Article(s):
Contributors: Dani Tran, Dr. Charles Schaefer, Dr. Charles Werth
Key Resource:
- Schaefer, C.E, Tran, D., Nguyen, D., Latta, D.E., Werth, C.J., 2025. Evaluating Mineral and In Situ Indicators of Abiotic Dechlorination in Clayey Soils[1]
Introduction
Cost-effective methods are needed to verify the occurrence of natural dechlorination processes and quantify their dechlorination rates in clays under ambient in situ conditions in order to reliably predict their long-term influence on plume longevity and mass discharge. However, accurately determining these rates is challenging due to slow reaction kinetics, the transient nature of transformation products, and the interplay of biotic and abiotic mechanisms within the clay matrix or at clay-sand interfaces. Tools capable of quantifying these reactions and assessing their role in mitigating plume persistence would be a significant aid for long-term site management.
For reductive abiotic dechlorination under anoxic conditions, a 1% hydrochloric acid (HCl) extraction of a sample of native clay coupled with X-ray diffraction (XRD) data can be used as a screening level tool to estimate reductive dechlorination rate constants. These rate constants can be inserted into fate and transport models such as REMChlor - MD[2][3] to quantify abiotic dechlorination impacts within clay aquitards on chlorinated solvent plumes. Thus, determination of the abiotic reductive dechlorination rate constant for a particular clayey soil can be readily utilized to provide a more accurate assessment of aquifer cleanup timeframes for groundwater plumes that are being sustained by contaminant back-diffusion.
Recommended Approach
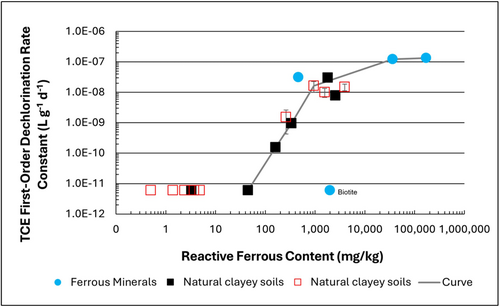
Figure 1: First-order rate constants for abiotic reductive dechlorination of TCE under anaerobic conditions. Circles are data from Schaefer
et al., 2021
[4], filled squares from Schaefer
et al., 2018
[5], and Schaefer
et al., 2017
[6], and open squares from Schaefer
et al., 2025
[1].
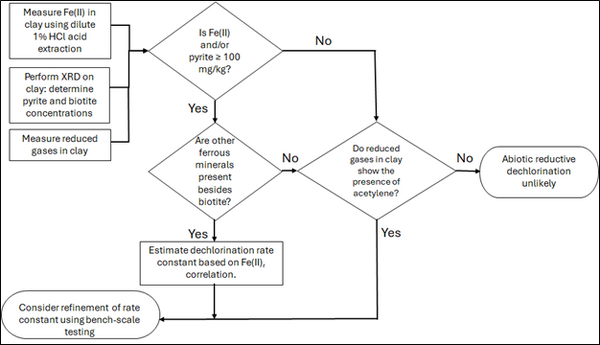
Figure 2: Flowchart diagram of field screening procedures
The recommended approach builds upon the methodology and findings of a recent study[1], emphasizing field-based and analytical techniques to quantify abiotic first-order reductive dechlorination rate constants for PCE and TCE in clayey soils under anoxic conditions. Key components of this evaluation are listed below:
- Zone Identification: The focus of the investigation should be to delineate clayey zones adjacent to hydraulically conductive zones.
- Ferrous Mineral Quantification: Assess ferrous mineral context in clay via 1% HCl extraction at ambient temperature over a 10-minute interval.
- Mineralogical Characterization: Conduct XRD analysis with the specific intent of identifying the presence of pyrite and biotite.
- Reduced Gas Analysis: Measurement of reduced gases such as acetylene, ethene, and ethane concentrations in clay samples. Gas-tight sampling devices (e.g., En Core® soil samplers by En Novative Technologies, Inc.) should be used to ensure sample integrity during collection and transport.
Clay samples should be collected within a few centimeters of the high-permeability interface, with optional additional sampling further inward. For mineralogical analysis, a defined interval may be collected and subsequently subsampled. To preserve sample integrity, exposure to air should be minimized during collection, transport, and handling. Homogenization should occur within an anaerobic chamber, and if subsamples are required for external analysis, they must be shipped in gas-tight, anaerobic containers.
Estimation of the abiotic reductive first-order rate constant for PCE and TCE is based on the “reactive” ferrous content in the clay. Reactive ferrous content (Fe(II)r) is estimated as shown in Equation 1:
- Equation 1: Fe(II)r = DA + XRDpyr - XRDbiotite
where DA is the ferrous content from the dilute acid (1% HCl) extraction, XRDpyr is the pyrite content from XRD analysis, and XRDbiotite is the biotite content from XRD analysis[1].
Abiotic dechlorination is unlikely to contribute to mitigating contaminant back-diffusion when reactive ferrous iron (Fe(II)r) concentrations are below 100 mg/kg (Figure 1). For Fe(II)r above 100 mg/kg, the first-order rate constant for PCE and TCE reductive dechlorination can be estimated using the correlation shown in Figure 1[5][7]. The rate constant exhibits a strong positive correlation with the logarithm of reactive Fe(II) content (Pearson’s r = 0.82), with a slope of 4.7 × 10⁻⁸ L g⁻¹ d⁻¹ (log mg kg⁻¹)⁻¹.
Figure 2 presents a decision flowchart designed to evaluate the significance and extent of abiotic reductive dechlorination. By applying Equation 1 to the dilute acid extractable Fe(II) plus measured mineral species data from clay samples, the reactive ferrous iron content (Fe(II)r) can be quantified, enabling a streamlined assessment of the extent to which abiotic processes are contributing to the mitigation of contaminant back-diffusion.
If Fe(II)r is ≥ 100 mg/kg, a first-order dechlorination rate constant can be estimated and subsequently used within a contaminant fate and transport model. However, if acetylene is detected in the clay, even with Fe(II)r less than 100 mg/kg, then bench-scale testing using methods similar to those described in a recent study[1] is recommended, as such results would likely be inconsistent with those shown in Figure 1, suggesting some other mechanism might be involved, or that the system mineralogy might be more complex than anticipated. Even if Fe(II)r ≥ 100 mg/kg, confirmatory bench-scale testing may be conducted for additional verification and to refine estimation of the abiotic dechlorination rate constant.
Summary and Recommendations
The approach outlined above is intended to serve as a generalized guide for practitioners and site managers to cost-effectively determine the extent to which beneficial abiotic reductive dechlorination reactions are likely occurring in low permeability (e.g., clayey) zones. This approach may be contraindicated if co-contaminants are present. It is currently unclear whether other classes of potentially reactive chemicals, such as trinitrotoluene (TNT) or chlorinated ethanes, could interact competitively with PCE and TCE.
In addition, it remains unclear how other classes of compounds such as per- and polyfluoroalkyl substances (PFAS) may interact or sorb with ferrous minerals and potentially inhibit abiotic dechlorination reactions. Coupling these recommended activities with conventional site investigation tasks would provide an opportunity to perform many of the up-front screening activities with minimal additional project costs. It is important to note that the guidance proposed herein pertains to particularly low permeability media. Sites with complex or varying lithology, where the mineralogy and/or redox conditions may vary, might require evaluation of multiple samples to provide appropriate site-wide information.
References
- ^ 1.0 1.1 1.2 1.3 1.4 Schaefer, C.E., Tran, D., Nguyen, D., Latta, D.E., Werth, C.J., 2025. Evaluating Mineral and In Situ Indicators of Abiotic Dechlorination in Clayey Soils. Groundwater Monitoring and Remediation, 45(2), pp. 31-39. doi: 10.1111/gwmr.12709
- ^ Falta, R., and Wang, W., 2017. A semi-analytical method for simulating matrix diffusion in numerical transport models. Journal of Contaminant Hydrology, 197, pp. 39-49. doi: 10.1016/j.jconhyd.2016.12.007 Open Access Manuscript
- ^ Kulkarni, P.R., Adamson, D.T., Popovic, J., Newell, C.J., 2022. Modeling a well-charactized perfluorooctane sulfate (PFOS) source and plume using the REMChlor-MD model to account for matrix diffusion. Journal of Contaminant Hydrology, 247, Article 103986. doi: 10.1016/j.jconhyd.2022.103986 Open Access Manuscript
- ^ Schaefer, C.E., Ho, P., Berns, E., Werth, C., 2021. Abiotic dechlorination in the presence of ferrous minerals. Journal of Contaminant Hydrology, 241, 103839. doi: 10.1016/j.jconhyd.2021.103839 Open Access Manuscript
- ^ 5.0 5.1 Schaefer, C.E., Ho, P., Berns, E., Werth, C., 2018. Mechanisms for abiotic dechlorination of trichloroethene by ferrous minerals under oxic and anoxic conditions in natural sediments. Environmental Science and Technology, 52(23), pp.13747-13755. doi: 10.1021/acs.est.8b04108
- ^ Schaefer, C.E., Ho., Gurr, C., Berns, E., Werth, C., 2017. Abiotic dechlorination of chlorinated ethenes in natural clayey soils: impacts of mineralogy and temperature. Journal of Contaminant Hydrology, 206, pp. 10-17. doi: 10.1016/j.jconhyd.2017.09.007 Open Access Manuscript
- ^ Borden, R.C., Cha, K.Y., 2021. Evaluating the impact of back diffusion on groundwater cleanup time. Journal of Contaminant Hydrology, 243, Article 103889. doi: 10.1016/j.jconhyd.2021 Open Access Manuscript
See Also

